
Chelation Characteristics
A chelate occurs whenever a metal is held suspended by two or more bonds from different atoms of the same molecule. Once the bonds are made, ring structures of atoms are formed by the surrounding molecule. Plants have been utilizing the principles of chelation since plants have been in existence. What is relatively new is the creation of mineral chelates in biocompatible forms that can supply highly bioavailable minerals to plants.
Chemical Properties of Chelates
In agricultural settings, the term chelate has often been misunderstood or misapplied in a general or catch-all fashion. Although chelation is a defined chemical term, the molecules (also called ligands) which bind to the minerals may vary widely in their physical, chemical, and nutritional properties. For this reason, it is not sufficient to refer to a mineral nutrient as a chelate, inferring that any one chelate is just like any other one. Among the different types of chelated molecules, nutritional quality depends on the type of the ligands and the proper chemistry of making chelates. The proposed ligand must have chemically reactive sites (moieties) capable of participating in the chelation process. The reactants must also be in the proper mole ratios for chemical combination to occur. There are additional considerations of spatial orientation. If the chelating moieties do not align properly for a feasible bond to form without straining the molecular structure, it will not form. If the ligand is too large, the probability of reactive moieties coming close enough for chelation to occur is remote.
The Necessity of Proper Ratios
The mole is a concept used in chemistry to assure that adequate numbers of combining molecules are present in the right proportions for the needed reaction. One mole is defined as one gram equivalent of the molecular weight of any compound or element. Since a mole of any compound is equivalent to its molecular weight in grams, the weight differences between different molecules are nullified and one mole of any compound has the same number of molecules in it regardless of how large or small the molecule is. Likewise, one mole of a single element has the same number of individual atoms as there are molecules in one mole of any other compound. Thus, components used to build larger molecules can be combined as equivalents of proper mole ratios, assuring that every part has its partner.
Figure 1: An Amino Acid Chelate
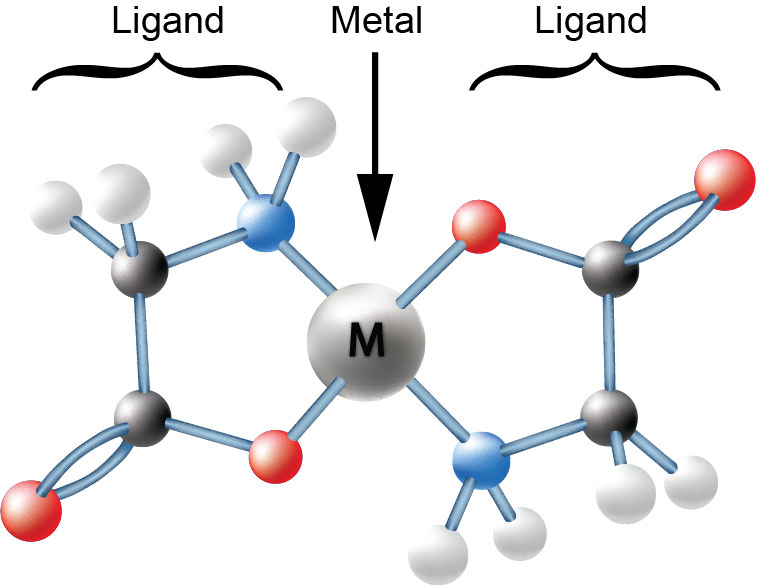
Measuring percentages of mineral atoms to combine with percentages of ligand molecules by weight are not molar ratios. Attempting to manufacture products using percentages or weight ratios to weigh out ingredients, results in products that can only be partially chelated, at best, or very remotely chelated, if at all. This is especially true if the proposed ligand is large with respect to the atomic weight of the metal being chelated. A crucial requirement of chelation, of course, presumes that the proper chemistry required to make a chelate is also present.
To successfully form a bioavailable chelate, it is also critical that the chelating ligand be small enough to form chelate bonds. Partially hydrolyzed protein is an example of a ligand that is too large to form true chelates.
Chelation of amino acids to specific metals is a precise process that does not just simply happen when one combines a few ingredients together. The process was developed and patented many years ago by the researchers at Albion. When it comes to amino acid chelation, Albion has written the book. For more information on how the Metalosate® products can help you in your business ventures, please contact your local Balchem Plant Nutrition representative.


